A new picture emerges on the origins of photosynthesis in a sun-loving bacteria

Every day, enough sunlight hits the Earth to power the planet many times over — if only we could more efficiently capture all the energy.
So scientists have looked at nature for inspiration to do it better.
By solving the heart of photosynthesis in a sun-loving, soil-dwelling bacterium, Raimund Fromme’s ASU research team has gained the most fundamental understanding of the inner workings of photosynthesis, and how this vital process differs between plants systems.
Their discovery may provide scientists with a brand-new template for laying the groundwork for organic-based solar panel design, known as “artificial leaves” for solar energy, or possible renewable biofuel applications.
The findings appear in today’s issue of Science.
Nature knows best
With today’s solar panels limited by their efficiency (currently, more than 80 percent of available solar energy is lost as heat), scientists have been trying to better understand the way photosynthetic plants and bacteria capture sunlight.
“Nature’s invention of photosynthesis is the single most important energy conversion process driving the biosphere, and photosynthesis forever changed the Earth’s atmosphere,” said Raimund Fromme, an associate research professor at the ASU Biodesign Institute’s Center for Applied Structural Biology and in the School of Molecular Sciences.
More than 3 billion years ago, our planet had an atmosphere without oxygen. At this time, nature figured out a way to capture the sunlight and convert it to food to take advantage of this everlasting energy source.
Life’s solar panels, which scientists call photosystems, are used by plants, algae and photosynthetic bacteria as an incredibly efficient system for capturing almost every available photon of light to grow and thrive, filling every nook and cranny on Earth.
Lightning in a bottle
Fromme is part of a large structural biology research group at ASU, who are steadily gaining a better understanding by taking pictures of the key proteins that work within the photosynthesis reaction centers to help turn light into energy.
“To truly and fully understand photosynthesis, one has to follow the process of converting light into chemical energy,” said Fromme. “This is one of the fastest chemical reactions ever studied, which is part of what makes it so hard to study and understand.”
The timescales of photosynthesis turn a bolt of lightning into a snail-like pace by comparison. Photosynthesis reactions occur at the scale of picoseconds, which is one-trillionth of a second. A picosecond is to one second as one second is to 37,000 years.
But the ASU structural biologists are using ever more powerful X-ray technology to one day catch up to the light by capturing freeze-frame images of crystallized proteins throughout the whole process.
In an effort to study photosynthesis, Fromme explored photosynthesis in its simplest form, in heliobacteria, which were first found in muddy soils near hot springs in far-flung locales like Iceland.
A murky life
The single-celled heliobacteria are simpler, yet fundamentally different than plants. For instance, during photosynthesis, instead of using water like plants, heliobacteria use hydrogen sulfide. They grow without oxygen, and after photosynthesis, give off a rotten-egg-smelling sulfur gas in place of oxygen.
Heliobacteria have used their unique place to successfully carve out their own ecological niche because they use a near-infrared wavelength of light for photosynthesis, which is perfect for low-light conditions found in places like Iceland or murky, muddy rice paddies. Plants simply can’t compete.
Scientists have wanted to understand how heliobacteria accomplish this.
Reaction to action
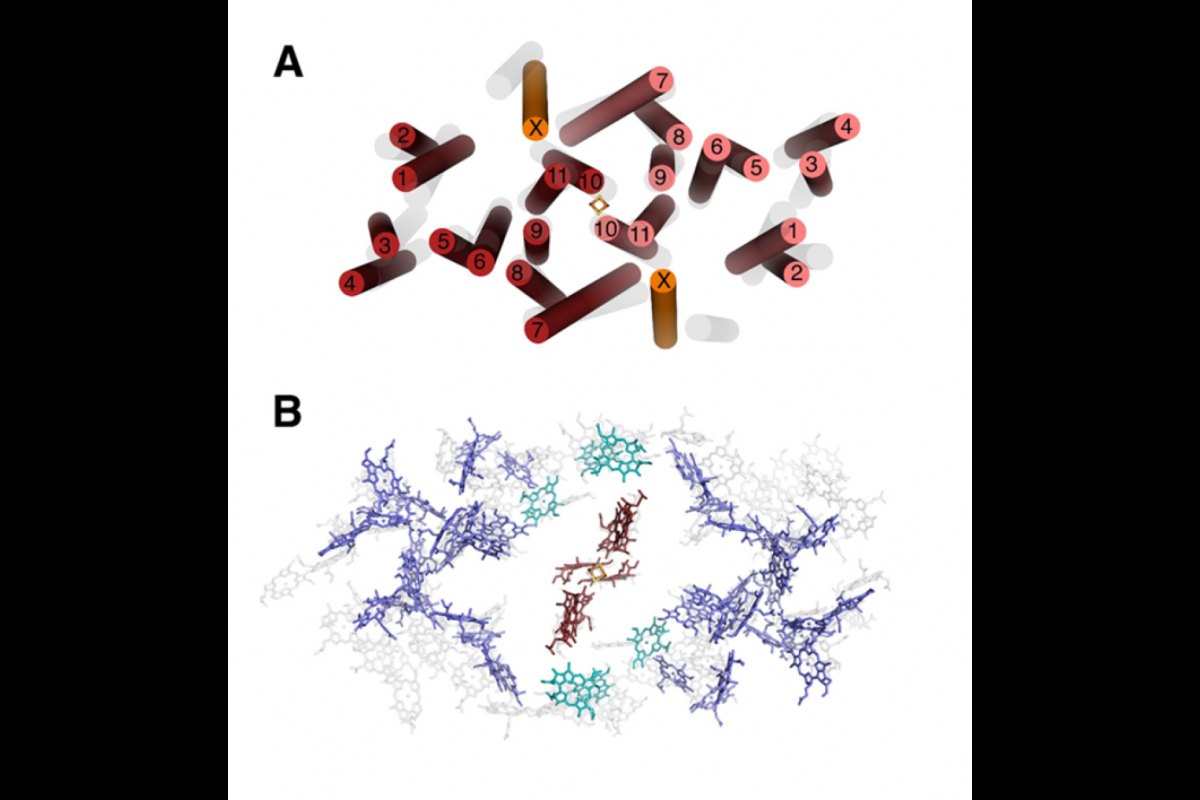
At the heart of photosynthesis is a reaction center (RC); it’s an elaborate complex of pigments and proteins that turn light into electrons to power the cell.
The heliobacteria RC has been proposed to be the closest thing alive to the earliest common ancestor of all photosynthetic reaction centers, when, around 3 billion years ago, the early Earth contained sulfur rich seas and little oxygen.
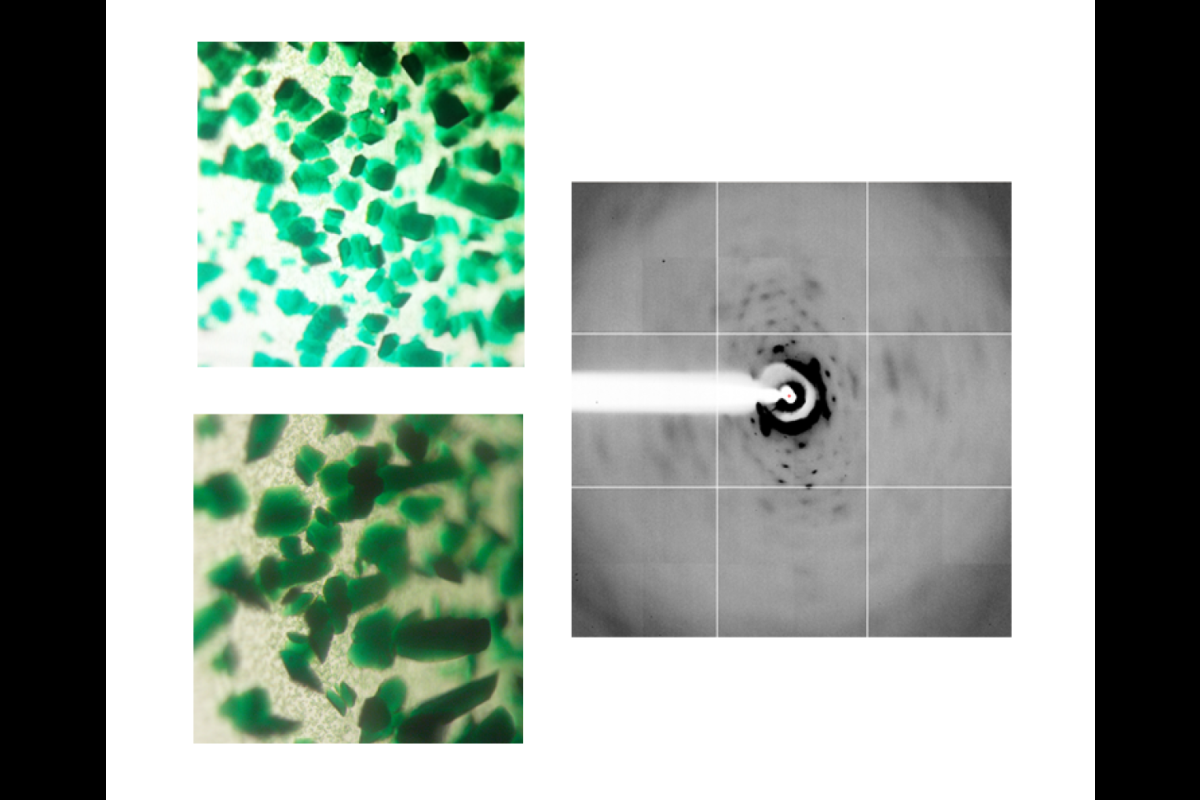
But successfully purifying an RC protein and growing crystals needed for X-ray experiments can be a lengthy, difficult process.
In particular, Fromme’s research project was started seven years ago as postdoctoral researcher Iosifina Sarrou first improved the preparation of the heliobacterial reaction center. After many initial trials of crystallization, an X-ray diffracting crystal charge was found.
“This is the moment a crystallographer is waiting for,” said Fromme, explaining the years it can take to grow the perfect protein crystal suitable for X-ray studies.
Two to tango
Short after these encouraging results, Christopher Gisriel joined the team and improved the diffraction quality of 2.2 angstroms to see the atomic details (a hydrogen atom is a single angstrom).
Still, the research team could not solve a crystallographic structure.
This hiatus took two years, until August 2016. Then, finally, a breakthrough came.
At this point “a thrilling discovery on unchartered territory began, as each new chlorophyll was cheered,” Fromme remembered, and “proved everyone’s initial prediction on the heliobacteria’s RC was wrong.”
Using X-ray light at the Advanced Light Source in Berkeley, California, and a beamline at Advanced Photon Source at Argonne National Lab, Illinois, Fromme’s group has now visualized the heliobacteria RCs for the very first time at near-atomic, 2.2-angstrom resolution (an angstrom is the width of a hydrogen atom).
They found an almost perfect symmetry in the heliobacter RC, which helps it gather every available photon of near infrared light.
The ancient origins of photosynthesis
Furthermore, with the explosion of DNA-sequencing technology, and with the potential ability to understand all of the genes and proteins across life, they also traced the evolution of the photosynthesis RCs.
They wanted to know: Could this simplest of reaction centers have spawned all others, leading to greater complexity over the eons?
In evolutionary terms, this means that the heliobacteria RC may have first come from a single gene.
“This structure preserves characteristics of the ancestral reaction center, providing insights into the evolution of photosynthesis,” explained Fromme’s colleague Kevin Redding, a researcher at ASU’s Center for Bioenergy and Photosynthesis. “From the new structures we have, it would certainly make sense for a compelling case.”
Then, the gene may have been duplicated to increase the evolutionary complexity.
“This likely occurred on at least three separate occasions, leading to the creation of all the different and more complex reactions center found in other photosynthetic bacteria and plants,” said Fromme.
The sun will rise again
Fromme’s group is excited about the potential of the new results. Such an understanding could one-day help research groups around the world build an artificial photosynthesis center.
ASU has long been a research leader in photosynthesis, going back to the 1970s. It was a natural fit for scientists drawn to the unique beauty of the Sonoran Desert that, with its 300-plus days of annual sunshine, is simply the best spot in the nation to capture solar energy.
And with ASU’s talent base, it may help spawn a local industry next-generation hybrid organic solar panels, possibly using the heliobacter or similar systems to boost light absorption and begin to boost solar energy efficiency or drive solar-based renewable biofuel technology.
After all, the sun will rise again tomorrow, awaiting more and more clever technologies scientists can dream up to capture the full potential of solar.
The team included Christopher Gisriel and Kevin E. Redding of ASU; Iosifina Sarrou of the Center for Free- Electron Laser Science, DESY; Bryan Ferlez and John H. Golbeck of Pennsylvania State University. This work was funded by the Division of Chemical Sciences, Geosciences, and Biosciences, Office of Basic Energy Sciences, of the U.S. Department of Energy through Grant (DE- SC0010575 to KR, RF, and JHG) and supported by X-ray crystallographic equipment and infrastructure provided by Petra Fromme of the Biodesign Center for Applied Structural Discovery at ASU.
More Science and technology

ASU-led space telescope is ready to fly
The Star Planet Activity Research CubeSat, or SPARCS, a small space telescope that will monitor the flares and sunspot activity of low-mass stars, has now passed its pre-shipment review by NASA.…

ASU at the heart of the state's revitalized microelectronics industry
A stronger local economy, more reliable technology, and a future where our computers and devices do the impossible: that’s the transformation ASU is driving through its microelectronics research…

Breakthrough copper alloy achieves unprecedented high-temperature performance
A team of researchers from Arizona State University, the U.S. Army Research Laboratory, Lehigh University and Louisiana State University has developed a groundbreaking high-temperature copper alloy…