ASU scientists utilize nonconventional techniques to make new materials

Christina Birkel, an inorganic chemist and assistant professor in the School of Molecular Sciences at ASU. Photo by Mary Zhu
Christina Birkel, an inorganic chemist and assistant professor in the School of Molecular Sciences at Arizona State University, is diligently working to create new materials that can be used for renewable energy, catalysts and permanent magnets.
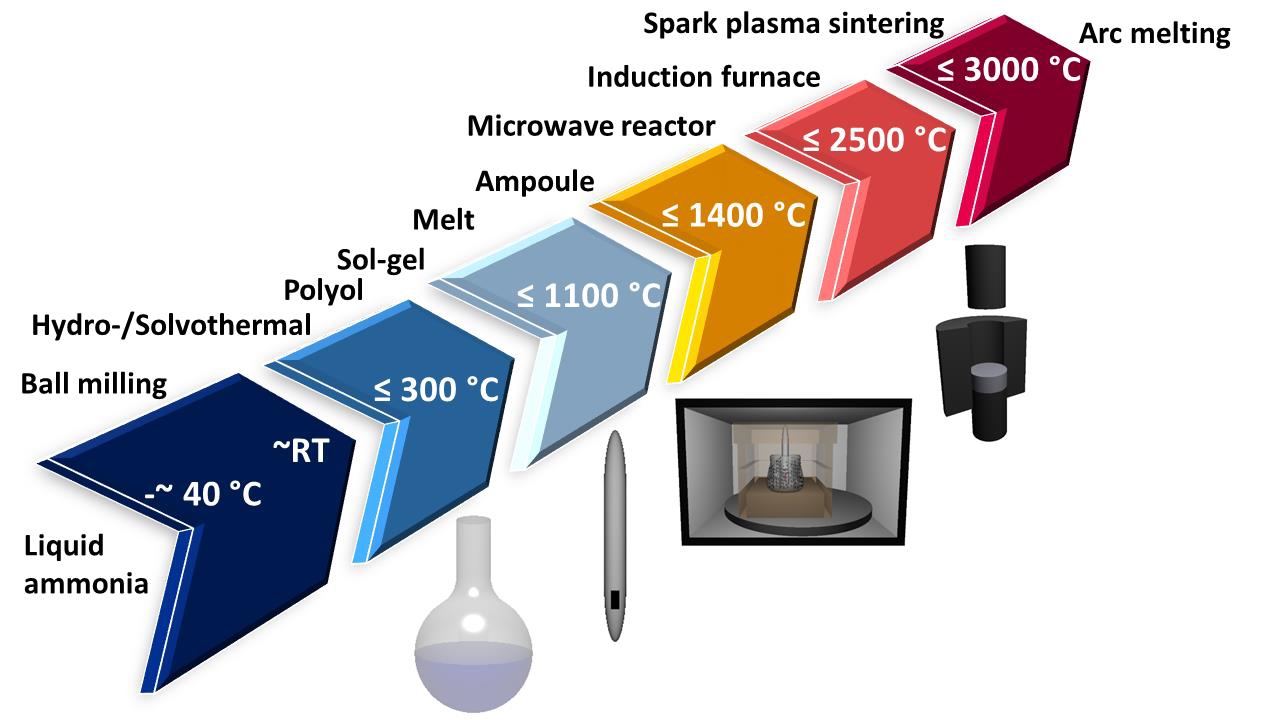
In this age of machine learning and data driven research a plethora of possible materials are being investigated, but synthesizing them in the lab, especially in the solid state, is often anything but straightforward.
"Compared to the highly developed methods for synthesis of molecular systems in solution, the rational synthesis of solid state materials is restricted to a limited number of useful techniques,” explained President’s Professor Ian Gould, interim director of the School of Molecular Sciences, which is part of The College of Liberal Arts and Sciences. “In this paper, Professor Birkel and her students nicely summarize two methods for solid-state synthesis that have already demonstrated potential for the preparation of new high value materials."
“My group is very active in the field of nonconventional synthesis techniques,” Birkel said. “We use microwave heating and spark plasma sintering to synthesize a lot of inorganic — i.e. not derived from living matter- compounds.”
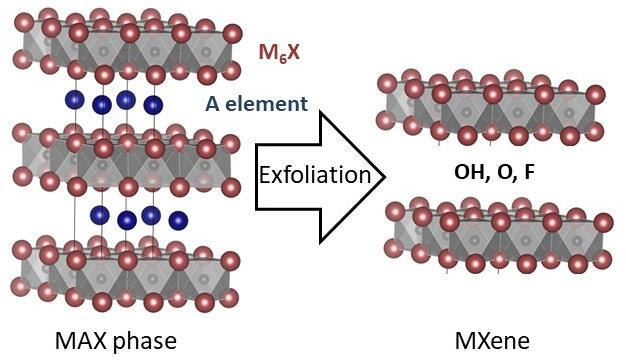
Birkel started out years ago working on oxides and metallic chalcogenides and recently her group has been very active in the field of carbides (so called MAX phases). Solid state microwave heating (using domestic and industrial ovens) is an excellent technique to synthesize carbides. Beautiful layered crystal structures are produced.
The Birkel group performs a lot of electron microscopy, X-ray diffraction and structural analysis on these materials. They are very special in that these materials can be exfoliated, that is, one layer of “A” elements in the structure (usually aluminum) can be etched out very selectively and you can end up with 2D materials (so called “MXenes”). These 2D materials are macroscopic in two dimensions and nanoscopic in the third. These materials have many potential applications including in the field of catalysis.
Birkel’s group is also active in the field of catalysis and is investigating the electrocatalytic properties of these materials, but at the same time they are asking a lot of very fundamental questions about how their synthesis techniques influence the structure and morphology of these compounds.
Where the 2D materials are concerned, it’s important to discover how the surface chemistry evolves during the exfoliation process. They are looking at how they can manipulate the surface chemistry and ultimately how that influences the catalytic properties.
More Science and technology

SpaceHACK highlights student solutions to environmental challenges, digital divide
By Adrianna Nine About 250 students from around the world convened online and at Arizona State University on March 22 for the…

New AI for a new era of discovery
As the legend goes, in 1665, Sir Isaac Newton sat in his garden at Woolsthorpe Manor in England and looked on as a lone apple…

ASU receives 3 awards for research critical to national security
Three researchers in the Ira A. Fulton Schools of Engineering at Arizona State University have received grant awards under the …